
How to produce green hydrogen?
Green hydrogen is produced by utilizing renewable energy sources such as solar, wind, and hydro. The process involves electrolysis, which splits water molecules into hydrogen and oxygen. During electrolysis, an electric current is passed through water, splitting it into hydrogen and oxygen.
The hydrogen produced through this process is considered “green” because it is generated using clean, renewable energy sources. The use of green hydrogen is expected to increase as the world moves towards more sustainable energy solutions and reduces its dependence on fossil fuels.
Hydrogen (H) is a powerful energy source due to its instability as an element, which leads to it easily bonding with other elements in nature. However, this also means that chemical reactions which produce a lot of heat are required to obtain H. This is where Lavoisier’s principle comes into play:
“Energy cannot be created or destroyed, only transformed.”
To produce hydrogen, a significant amount of energy is needed.
Types of hydrogen according to its origin
So far, H has been produced for industrial purposes such as hydrotreatment and fertilizer production, but it has been produced using methods that result in high levels of pollutants, mainly CO2.
- Gray hydrogen is traditionally produced using fossil fuels, particularly natural gas, which results in high levels of CO2 emissions. Despite this, it remains the most commonly used form of hydrogen due to its low cost.
- Blue hydrogen is produced using fossil energy, similar to gray hydrogen. However, the CO2 emissions generated during production are reduced through the use of CO2 capture and storage techniques. This makes blue hydrogen less polluting than gray hydrogen, although it is not a completely clean source of energy as some emissions still remain.
- Green hydrogen is produced through environmentally friendly methods that result in zero emissions. This type of hydrogen is generated mainly through the electrolysis of water and requires electricity from renewable sources like solar and wind power. This makes it the cleanest and most sustainable form of hydrogen available.
Although green form is considered the ideal form of hydrogen to support renewable energies in a new energy model, its production cost remains high and the technology is not yet fully developed.
New studies and new techniques
As issues with lithium batteries for energy storage emerge, interest in H as a clean energy source is growing. The production of solar batteries results in significant harm to the planet and political disputes, leading to increasing criticism of electromobility. This has led to a rise in the number of people opposing the use of lithium batteries.
As a result of these challenges, researchers are continuously exploring ways to make green hydrogen production more efficient and cost-effective. One example of this is the development of polymer membranes for electrolytes and advanced simulation and control techniques for the production process.
Green hydrogen production process

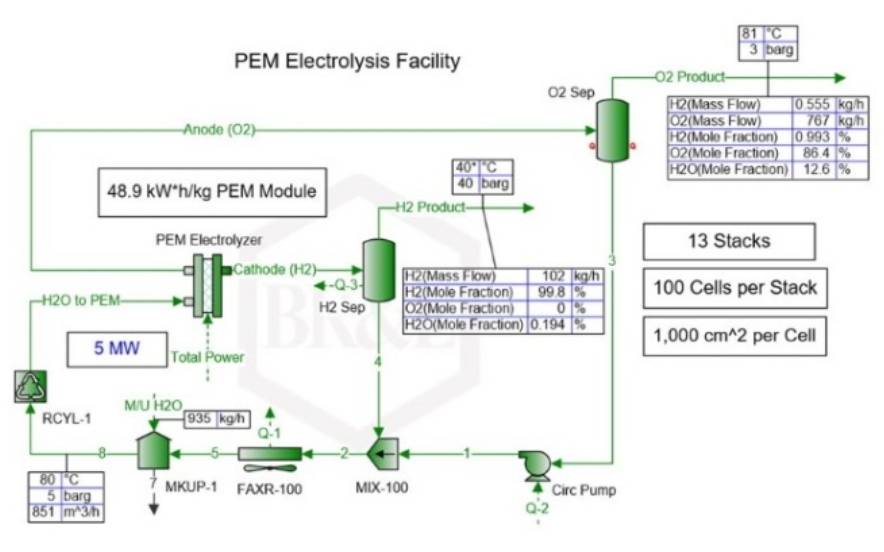
The production of renewable H involves the electrolysis of water, where an electrical current is applied to break down the water molecule into hydrogen and oxygen. A device capable of injecting water and applying the necessary electrical current is necessary to carry out this process.
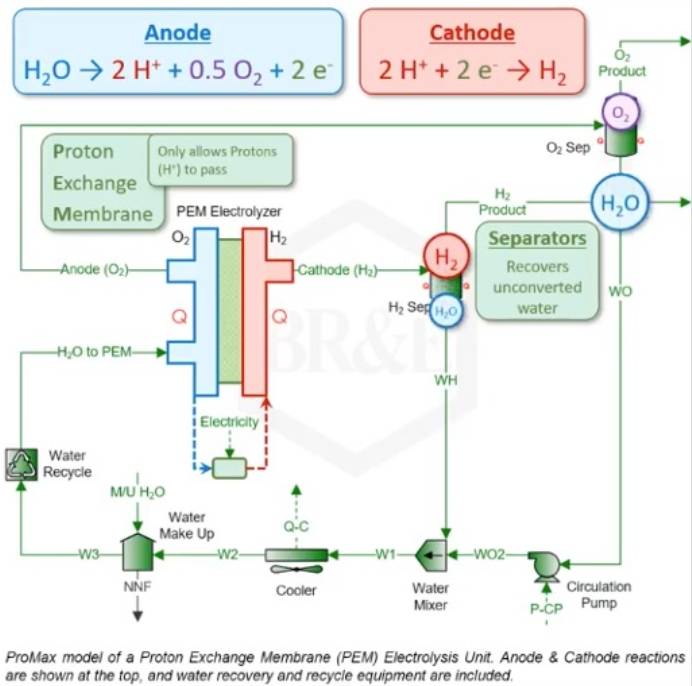
The production of green hydrogen through electrolysis requires an electrochemical cell consisting of a cathode and anode, with a proton exchange membrane (PEM) in between. The PEM is a special polymer membrane that separates the particles produced from the breakdown of the water molecule. This membrane acts as a filter and ensures the physical separation of hydrogen and oxygen.
Thus, each basic element of the electrolytic cell has an important function:
- The anode acts as the positive electrode during the electrolysis process, facilitating the flow of electric current. Through the anode, water is oxidized into H ions (H⁺), oxygen (O₂), and electrons (e-). Iridium/Ruthenium is commonly used as the material for the anode.
- The cathode, which is the negative pole, receives the H⁺ ions, which react with electrons (e-) to form hydrogen gas (H₂). This cathode is typically made of Platinum or coated with Platinum to improve its conductivity and catalytic properties. The cathode also helps to prevent the reverse reaction, in which hydrogen gas reacts with oxygen to form water. The end result of the electrolysis process is hydrogen and oxygen gases.
- The electricity for the electrolysis process is supplied by wind or solar photovoltaic power plants, which are typically located near the green hydrogen plant. This ensures a consistent supply of electricity for moving the electrons from the anode to the cathode.
- At the cathode, protons and electrons come together to form hidrogen molecules. The cathode is typically made of platinum and palladium.
Separators
The end product of the PEM electrolyzer is H on the cathode side and oxygen on the anode side, however both mixtures also contain residual water. Hence, the mixtures of hydrogen and water, as well as oxygen and water, must be separated using separators.
The mixtures of water and hydrogen and water with oxygen are then sent to separators where the water is separated from the hydrogen and oxygen flows. The separated water is cooled and recycled back to the PEM electrolyzer, along with a fresh water supply to maintain the required processing amount.
Renewable Hydrogen Uses

The use of H2-fuel cells for vehicles and trains, as well as for domestic boilers, has been explored, but widespread adoption for home use has yet to be achieved.
Additionally, the cost of producing green hydrogen is still relatively high, making it an expensive option for widespread consumer use. The technology also needs to be further developed and improved to make it a practical solution for the mass market. Despite these challenges, the potential benefits of green hydrogen are significant, and it is an area that continues to attract interest and investment from various sectors.
Additionally, the cost of setting up a H2-infrastructure for domestic use is currently very high, making it difficult for the average person to have access to it. The existing alternatives, such as solar panels or heat pumps, provide a more cost-effective solution for home energy use. Until green hydrogen production becomes more affordable and accessible, it is unlikely that it will become a widely used energy source in homes.
Another challenge to the widespread use of hydrogen in homes is the cost of its distribution. To make H2 available to households, the existing gas pipeline network would need to be adapted, and new pipelines would have to be installed within cities. These modifications require significant time and investment.

